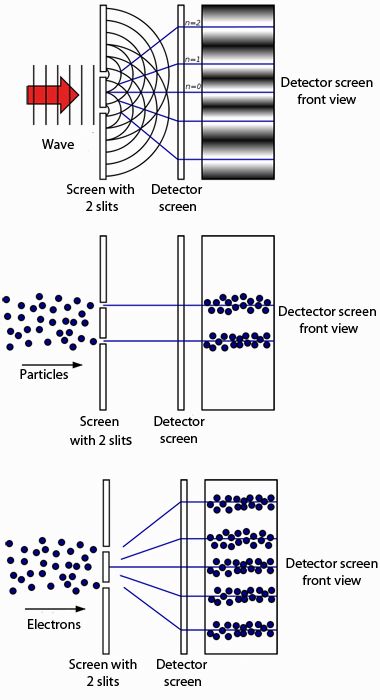
This was not an unsound proposal considering that macroscopic oscillators operate similarly when studying five simple harmonic oscillators of equal amplitude but different frequency, the oscillator with the highest frequency possesses the highest energy (though this relationship is not linear like Planck's). In 1900, Max Planck hypothesized that the frequency of light emitted by the black body depended on the frequency of the oscillator that emitted it, and the energy of these oscillators increased linearly with frequency (according E = hf where h is Planck's constant and f is the frequency). This became known as the ultraviolet catastrophe. This became clear when plotting the Rayleigh–Jeans law, which, while correctly predicting the intensity of long wavelength emissions, predicted infinite total energy as the intensity diverges to infinity for short wavelengths. But a problem quickly arose if each mode received an equal partition of energy, the short wavelength modes would consume all the energy. Since the equipartition theorem worked so well in describing the vibrational modes of the thermal object itself, it was natural to assume that it would perform equally well in describing the radiative emission of such objects. This became known as the black body problem. Since light was known to be waves of electromagnetism, physicists hoped to describe this emission via classical laws. That thermal objects emit light had been long known. But applying the same reasoning to the electromagnetic emission of such a thermal object was not so successful. The equipartition theorem of classical mechanics, the basis of all classical thermodynamic theories, stated that an object's energy is partitioned equally among the object's vibrational modes. Einstein later proposed that electromagnetic radiation itself is quantized, not the energy of radiating atoms.īlack-body radiation, the emission of electromagnetic energy due to an object's heat, could not be explained from classical arguments alone. atoms of the black body that emit radiation. To accomplish this, Planck had to make a mathematical assumption of quantized energy of the oscillators, i.e. In 1901, Max Planck published an analysis that succeeded in reproducing the observed spectrum of light emitted by a glowing object. History Classical particle and wave theories of light Turning the reasoning around, ordinary quantum mechanics can be deduced as a specialized consequence of quantum field theory. the electric and magnetic field strengths of Maxwell) are replaced by an entirely new kind of field value, as considered in quantum field theory. He saw it in what is called second quantization, which generates an entirely new concept of fields that exist in ordinary space-time, causality still being visualizable. He saw the duality as present for all quantic entities, but not quite in the usual quantum mechanical account considered by Bohr. Werner Heisenberg considered the question further. Bohr regarded renunciation of the cause-effect relation, or complementarity, of the space-time picture, as essential to the quantum mechanical account. He saw such duality as one aspect of the concept of complementarity. A given kind of quantum object will exhibit sometimes wave, sometimes particle, character, in respectively different physical settings. Īlthough the use of the wave–particle duality has worked well in physics, the meaning or interpretation has not been satisfactorily resolved see interpretations of quantum mechanics.īohr regarded the "duality paradox" as a fundamental or metaphysical fact of nature. For macroscopic particles, because of their extremely short wavelengths, wave properties usually cannot be detected. This phenomenon has been verified not only for elementary particles, but also for compound particles like atoms and even molecules. Through the work of Max Planck, Albert Einstein, Louis de Broglie, Arthur Compton, Niels Bohr, Erwin Schrödinger and many others, current scientific theory holds that all particles exhibit a wave nature and vice versa. We have two contradictory pictures of reality separately neither of them fully explains the phenomena of light, but together they do. We are faced with a new kind of difficulty. It seems as though we must use sometimes the one theory and sometimes the other, while at times we may use either. It expresses the inability of the classical concepts "particle" or "wave" to fully describe the behaviour of quantum-scale objects. 
Wave–particle duality is the concept in quantum mechanics that every particle or quantum entity may be described as either a particle or a wave.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
